
The numbers and titles may be referred to on nonconformity reports prepared by the auditor. The bold numerical typescripts used in the first two columns of the checklist with titles indicate the Requirements. Evidence for visible top management commitment and quality management action must be looked for. 
Here is a list of the standards referenced in ISO bibliography ISO 9001:2000, Quality management systems Requirements ISO 10012, Measurement management systems Requirements for measurement processes and measuring equipment ISO 11134:1994, Sterilization of health care products Requirements for validation and routine control Industrial moist heat sterilization ISO 11135:1994, Medical devices Validation and routine control of ethylene oxide sterilization (Corrigendum 1 published 1994) ISO 11137:1995, Sterilization of health care products Requirements for validation and routine control Radiation sterilization (Corrigendum 1 published 1995 Amendment 1 published 2001) ISO 13641:2002 Part 1 General test & ISO 13641:2002 Part 2 Test for low biomass concentrations, Elimination or reduction of risk of infection related to in vitro diagnostic medical devices ISO 13683:1997, Sterilization of health care products Requirement for validation and routine control of moist heat sterilization in health care facilities ISO :2003, Clinical investigation of medical devices for human subjects Part 1: General requirements ISO :2003, Clinical investigation of medical devices for human subjects Part 2: Clinical investigation plans ISO 14160:1998, Sterilization of medical devices Validation and routine control of sterilization of single-use medical devices incorporating materials of animal origin by liquid chemical sterilants ISO 14937:2000, Sterilization of health care products General requirements for characterization of a sterilizing agent and the development, validation and routine control of a sterilizing agent ISO/TR 14969: 1), Medical devices Quality management systems Guidance on the application of ISO 13485:201:2002, Guidelines for quality and/or environmental management systems auditingĢ Auditors must be careful and thoughtful prior to establishing a deficiency against a requirement. ISO 14971:2007 Medical devices Application of risk management to medical devices ISO Guide Risk management - Vocabulary. 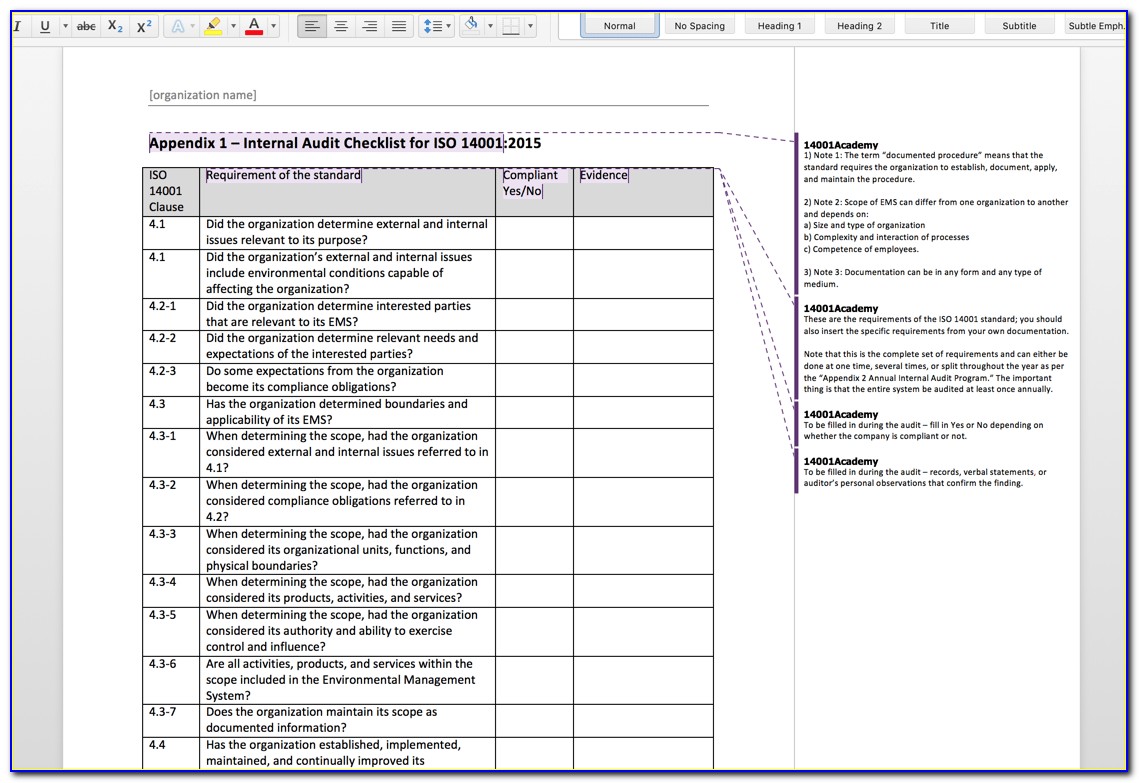
Risk Management is a requirement-product Realization clause 7.1 o See guidance standards. 5: INTERNAL AUDIT CHECKLIST = + Requirements in addition to ISO 9001 are Highlighted in yellow in this document Throughout this document, you will find the following assistance: Links to supporting information are underlined blue text Links to buy Standards directly from the source are Underlined Bold Red text Here are some resources: Comparison between ISO 9001-and-ISO Buy copies of the ISO13485 standard to pinpoint the areas that need attention.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
